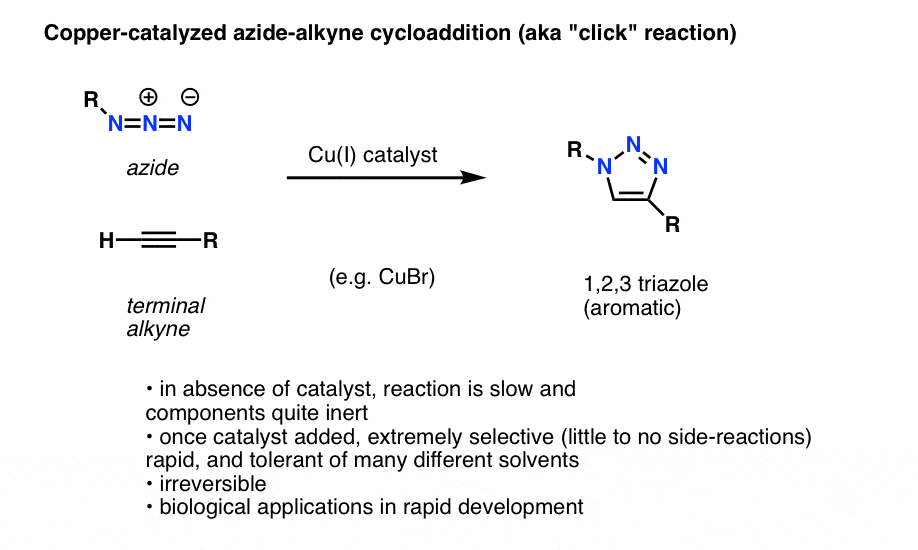
Reaction scheme of the ring-opening process in presence of sodium azide. | Download Scientific Diagram

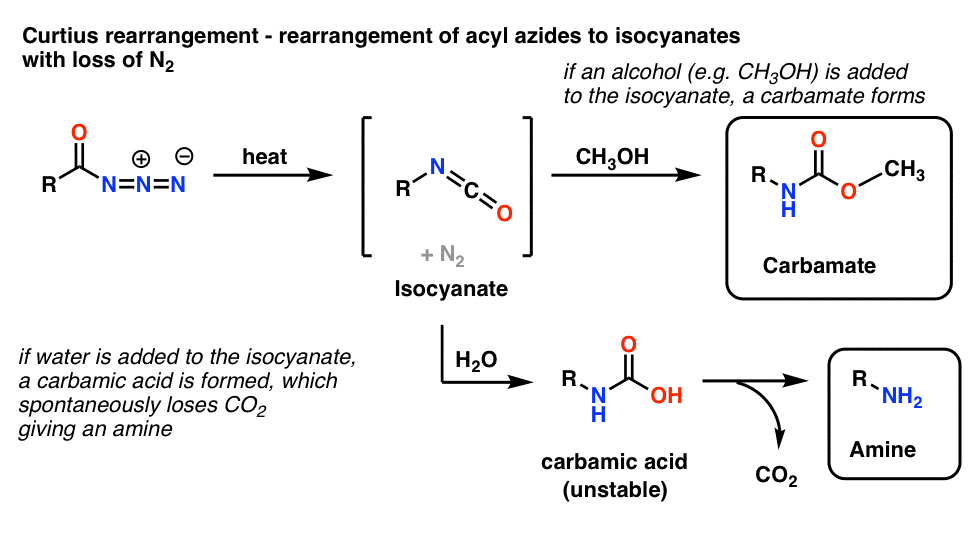
Synthesis of 1‐Pyrroline by Denitrogenative Ring Expansion of Cyclobutyl Azides under Thermal Conditions - Miki - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library
![Tandem Epoxide or Aziridine Ring Opening by Azide/Copper Catalyzed [3+2] Cycloaddition: Efficient Synthesis of 1,2,3-Triazolo β-Hydroxy or β-Tosylamino Functionality Motif | The Journal of Organic Chemistry Tandem Epoxide or Aziridine Ring Opening by Azide/Copper Catalyzed [3+2] Cycloaddition: Efficient Synthesis of 1,2,3-Triazolo β-Hydroxy or β-Tosylamino Functionality Motif | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo701724f/asset/images/jo701724f.social.jpeg_v03)
Tandem Epoxide or Aziridine Ring Opening by Azide/Copper Catalyzed [3+2] Cycloaddition: Efficient Synthesis of 1,2,3-Triazolo β-Hydroxy or β-Tosylamino Functionality Motif | The Journal of Organic Chemistry

Ring Opening of Epoxides and Aziridines with Sodium Azide using Oxone® in Aqueous Acetonitrile: A Highly Regioselective Azidolysis Reaction
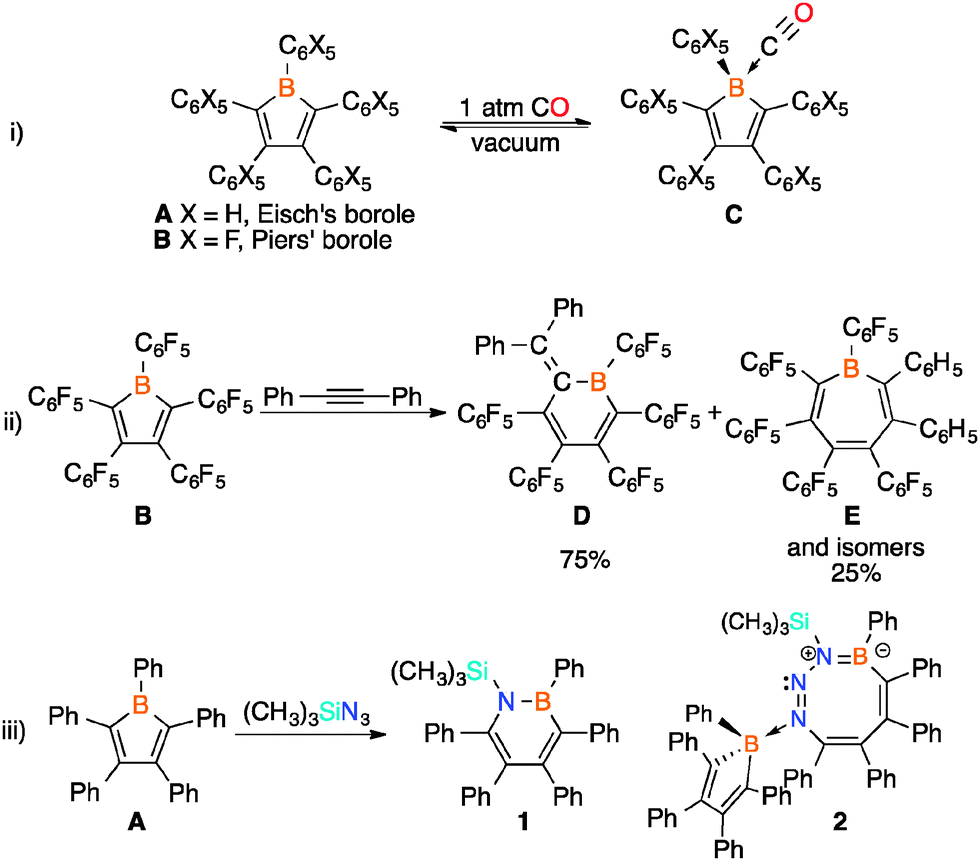
Investigating the ring expansion reaction of pentaphenylborole and an azide - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC04864D

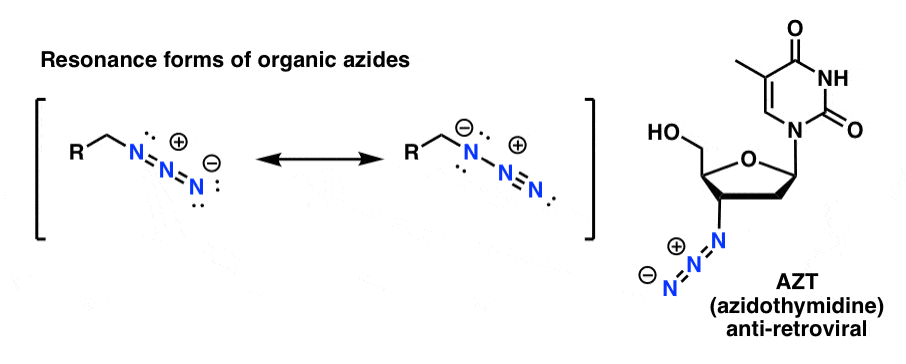
Asymmetric Schmidt Reaction of Hydroxyalkyl Azides with Ketones | Journal of the American Chemical Society
![One pot 'click' reactions: tandem enantioselective biocatalytic epoxide ring opening and [3+2] azide alkyne cycloaddition - Chemical Communications (RSC Publishing) One pot 'click' reactions: tandem enantioselective biocatalytic epoxide ring opening and [3+2] azide alkyne cycloaddition - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/B919434G)
One pot 'click' reactions: tandem enantioselective biocatalytic epoxide ring opening and [3+2] azide alkyne cycloaddition - Chemical Communications (RSC Publishing)

The trans–cis and the azide–tetrazole ring-chain isomerization of 2-azido-1,3-azoles: Quantum chemical study - ScienceDirect

Copper-Catalyzed Azide–Alkyne Cycloaddition of Hydrazoic Acid Formed In Situ from Sodium Azide Affords 4-Monosubstituted-1,2,3-Triazoles | The Journal of Organic Chemistry






![Synthonix, Inc > Azides > 2-Thiophenecarbonyl azide - [T73481] Synthonix, Inc > Azides > 2-Thiophenecarbonyl azide - [T73481]](https://synthonix.com/images/product/73481.png)